We use cookies and other similar technologies to improve your browsing experience and the functionality of our site. By clicking "Accept All Cookies", you consent to the storing on your device of all the technologies described in our Cookie Policy. Your current cookie settings can be changed at any time by clicking "Cookie Preferences". We also urge you to read our Privacy Policy and Terms of Use. Using the site equals consent to the use of cookies.

We regret to inform you that the Emanuel Berg online store does not ship to the United States of America and Canada.
We regret to inform you that the Emanuel Berg online store does not ship to the United States of America and Canada.
THE FIRST SHIRT THAT PROTECTS YOU FROM THE VIRUS

VIROFORMULA
ANTIVIRAL PROTECTION MAY BECOME A REGULAR FEATURE OF CLOTHING IN THE COMING YEARS.
EMANUEL BERG UNDERSTANDS THE DEMAND FOR THIS KIND OF PRODUCT AND HEREBY PRESENTS A SPECIAL OFFER
What is it?
An innovative fabric that stems from the world-renowned mill of Albini. Based on their close collaboration with HeiQ, a Swiss laboratory, they have developed a textile barrier against viruses and bacteria which can easily be integrated into your everyday life.
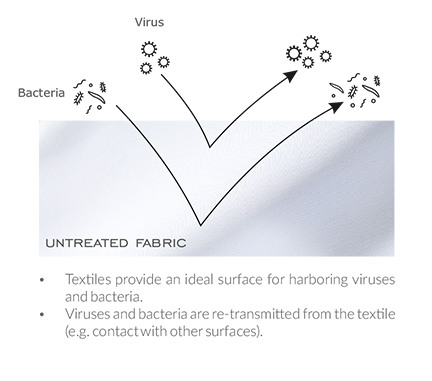
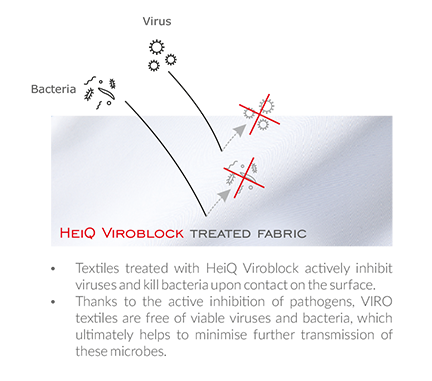
How it works?
The technology works, based on silverions and liposomes, which attract and attack viral and bacterial cells that come in contact with the surface of your shirt or face mask.
How to care for your Viro shirt?
The activity of VIRO lasts up to 30 washes in the washing machine, at a maximum of 40°C. The formula is dermatologically tested as non-irritable and has been deemed as an environmentally sustainable product.
Disclaimer:
This product is treated with a biocide to preserve it from the settlement of microorganisms, such as viruses and bacteria, on its surface. It contains silver (nano). WARNING: The biocidal properes of this garment persist up to a maximum of 30 washes (max. 40ºC). Air-dry the garment. Avoid unnecessary washing to limit the dispersion of biocide in the environment. This product has no primary biocidal function and does not replace the ordinary norms of prevention of bacterial or viral infections. The product we offer is not a medical device and has not been cerfied for medical use or for any other similar function
VIRO is our new capsule of shirts and face masks, made of VIROFORMULA fabrics which have been produced byAlbini in collaboraon with HeiQ. They contain a biocide, or active substances (silver chloride and tanium dioxide), which preserve our VIRO items from the settlement of microbes on its surface. As a result, the spread of bacteria and viruses is reduced.
The biocide is authorised under EU Regulation 2012/528 (“BPR Regulation”) and is provided with cerfication N-91819, issued by the manufacturer of the biocide treatment - HeiQ Materials.
The treatment with HeiQ's Viroblock technology gives our VIRO products secondary biocidal properes, thus classifying them as “Treated Arcle”, pursuant to art. 3, paragraph 1 leer L) ofthe BPR Regulation, according to which: a Treated Article is “any substance, mixture or article which has been treated with, or intentionally incorporates, one or more biocidal products” as long as it does not have a primary biocidal function.











